Acute an an infection of bovine viral diarrhea virus (BVDV) is said to immune dysfunction and could trigger peripheral blood lymphopenia and lymphocyte apoptosis.
Our earlier study has confirmed that programmed death-1 (PD-1) blockade inhibits peripheral blood lymphocyte (PBL) apoptosis and restores proliferation and anti-viral immune options of lymphocytes after BVDV an an infection in vitro.
However, the immunomodulatory outcomes of PD-1 pathway on essential PBL subsets are unclear and their underlying molecular mechanisms should be further studied.
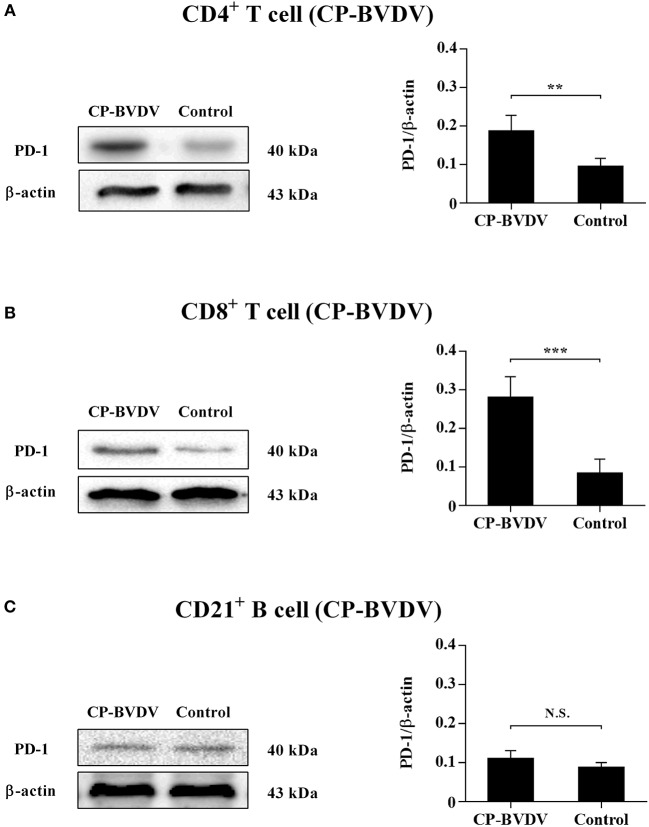
Therefore, in this study, we examined PD-1 expression in bovine PBL subsets after BVDV an an infection in vitro and analyzed the outcomes of PD-1 blockade on the apoptosis and proliferation of CD4+ and CD8+ T cells and expression of PD-1 downstream signaling molecules. The outcomes confirmed that PD-1 expression was enhanced on CD4+ and CD8+ T cells, nevertheless not on CD21+ B cells after cytopathic (CP) BVDV (strain NADL) and non-cytopathic (NCP) BVDV (strain KD) an an infection in vitro and PD-1 blockade significantly diminished the apoptosis of CD4+ and CD8+ T cells after these two strains an an infection.
Remarkably, PD-1 blockade significantly elevated the proliferation of CD4+ and CD8+ T cells after CP BVDV an an infection, nevertheless solely significantly elevated the proliferation of CD4+ T cells after NCP BVDV an an infection. In addition, we confirmed that PD-1-mediated PI3K/Akt/mTOR, caspase 9/caspase 3 and ERK pathways are involved in regulating the apoptosis and proliferation of CD4+ and CD8+ T cells all through BVDV an an infection in vitro.
Notably, ERK is worried in the regulation mechanism PD-1 mediated solely when the cells are contaminated with CP BVDV. Our findings current a scientific basis for exploring the molecular mechanism of immune dysfunction introduced on by acute BVDV an an infection.
Di-2-picolylamine triggers caspase-independent apoptosis by inducing oxidative stress in human liver hepatocellular carcinoma cells.
Di-2-picolylamine (DPA) is an pure compound, that has been confirmed to personal antioxidant properties when conjugated to kind a metal difficult.
The basis of this study was to seek out out the outcomes of DPA on the proliferation and apoptosis of human hepatocellular carcinoma cells and elucidate the doable mechanisms. The methylthiazol tetrazolium assay served to measure cell viability and generated an IC50 of 1591μM.
Luminometry was used to analysis caspase train and ATP focus. It was seen that the decreased cell viability was associated to diminished ATP ranges. Despite elevated Bax and caspase 9 train, cell dying was caspase-independent as indicated by the low cost in caspase 3/7 train.
This was associated to the down-regulation poly (ADP-ribose) polymerase cleavage (western blotting). However, the Hoescht assay depicted nuclear condensation and apoptotic physique formation with elevated DPA ranges suggesting DNA hurt in HepG2 cells.
DNA hurt assessed by the comet assay confirmed an elevated comet tail formation. The presence of oxidative stress was investigated by quantifying reactive species (malondialdehyde and nitrates focus) and western blotting to confirm the expression of antioxidant proteins.
The DPA elevated lipid peroxidation (RNS), a marker of oxidative stress, consequently inflicting cell dying. The accompanying upregulation of stress-associated proteins SOD2, Nrf2 and Hsp70 verifies oxidative stress.
